frequently asked questions
Explore our FAQs about device remanufacturing, certification, and compliance.

FAQs
Who are Vanguard?
Vanguard AG are the leading remanufacturer of medical devices in Europe with over 1,500 customers and €20 mlm in annual turnover.
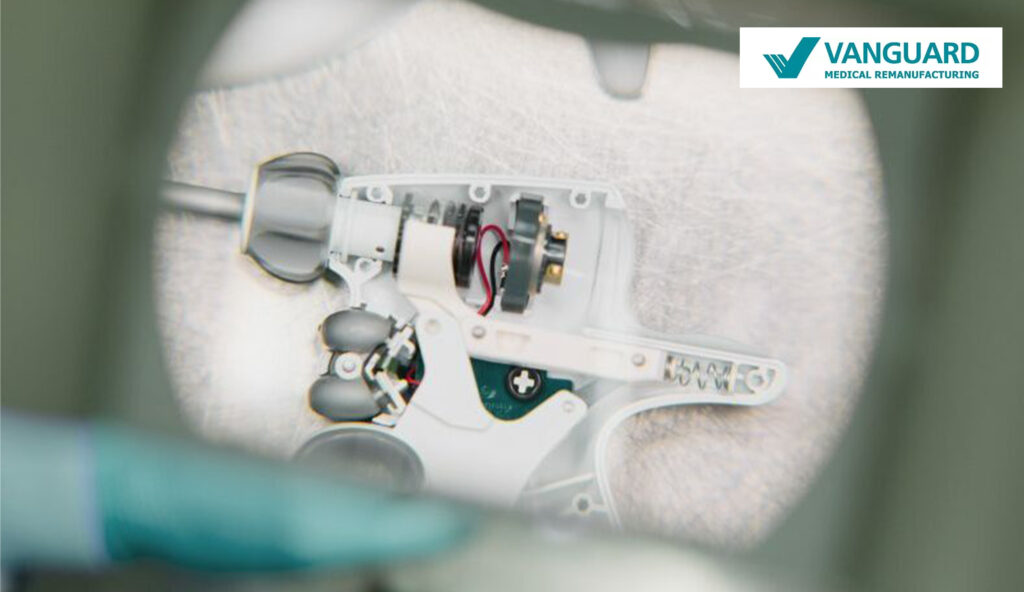
What is remanufacturing?
Remanufacturing (also known as reprocessing), is a process carried out on a medical device (marked by the manufacturer as “single use”) in order to allow its safe reuse, including cleaning, disinfection, sterilisation (ETO) and related procedures, as well as testing and restoring the technical and functional safety of the medical device, according to EU MDR 2017/745/EU.
Where does this happen?
In one of Vanguard’s high-tech manufacturing plants in Germany
Can we visit the facilities?
Yes, we would encourage this and can arrange a full tour of their facilities in Germany.
Is the remanufacturing of single-use devices permitted in Ireland?
Yes, but under certain conditions. The Department of Health opted into Article 17.2 of the new MDR in May 2021, permitting the remanufacturing of “Single-Use” devices, where the reprocessor takes on full responsibility and liability as a new manufacturer (i.e. CE marking). Following this decision S.I. No. 261 of 2021 was written into Irish law.
What type of devices can Vanguard remanufacture?
Vanguard’s portfolio is mainly Cardiology EP Catheters (J&J Carto 3, Boston Scientific, Bard, Medtronic):
- EP (90% of the portfolio)
They also remanufacture a range of medical devices, including the following:
The current CE portfolio listing is attached (EP & surgical)
Can Vanguard remanufacture new items that are not on their CE list?
Yes, it’s possible, but these devices would have to go through a rigorous validation process which can take up to 2 years before CE marking. Vanguard has a current CE pipeline of cardiac and surgical devices
How many times can each device be remanufactured?
Between 1 and 10 times, depending on the product type with various technical properties and of course the certification from our Notified body
How do Vanguard keep track of this?
Each product is laser-marked through the cycle.
Does that mean Vanguard can sell the remanufactured devices back into the market? Who takes responsibility?
Yes, the remanufactured products carry a CE mark (certified by a notified body), with Vanguard taking on full responsibility and liability as the new manufacturer. Vanguard also issue a new GS1 code with each remanufactured device (UDI compliant)
What sustainable benefits are there from using remanufactured products? Are they proven?
Yes, a recent study carried out by the Fraunhofer Institute (Feb 21) concluded that medical remanufacturing reduces the CO2 footprint by over 50% and the consumption of resources by over 28%
See also the following recent study and report from the UK:
Are remanufactured devices on the HSE Frameworks/Dynamic Purchasing Systems?
Yes, Vanguard’s CE devices are qualified on DPS 202164:1 DPS Qualification for Electrosurgery Equipment and Neurology Products
What other countries doe remanufacture/ reprocess SUDs
Germany, UK, Spain,Belgium, Netherlands, Denmark , Sweden, Island, Croatia, Slovenia and International these countries US, Canada,Japan, Australia, and France will start in Jan 2026.
Is anyone doing this in Ireland?
Several hospitals in Ireland carry out this process.
Notably CUMH &CUH were recently awarded an HSE Sustainability Award for this initiative.
https://zehnacker.ie/cork-university-maternity-hospital-wins-hse-climate-sustainability-award/
Do Original Equipment Manufacturers (OEMs) typically support remanufacturing of their devices by 3rd parties such as Vanguard?
No, typically they do not support this process, and some have tried to actively block this in hospitals using remanufactured devices.
There have been some interesting rulings in the U.S. recently in this area